Menu
Tendencias Periodicas y Vinculacion
Chemistry Textbook crayon color labels Elements of Innovation Project Outline
Chemistry Textbook crayon color labels Elements of Innovation Project Outline
Periodic Table Groups Valence Electrons Periodic Trends: Ionization Energy
Other Trends Bonding: Ionic, Covalent and Metallic
http://tinyurl.com/Barnes-Nerds
Other Trends Bonding: Ionic, Covalent and Metallic
http://tinyurl.com/Barnes-Nerds
|
Grupos de la Tabla Periodica: Big Hero 6 Lab
thanks to NASA's Energy from the Sun Click on pics to see student samples: |

Entry Letter Fall 2014.
Driving question: How does the arrangement of the elements in the periodic table relate to their properties and uses in innovative technologies?
Read the Advanced Materials article and discuss two types of innovative materials you find. Describe their uses and why they are considered innovative.
Featuring the We Care Solar Briefcase
Read your Chemistry Text
- sec 6.1, Organizing the Elements and complete 6.1 assessment.
- sec 6.2, Classifying the Elements and complete 6.2 assessment.
- share on a google doc titled: Ch6 Organizing & Classifying Elements (your name)
- Start to read sec 6.3, Periodic Trends
Caracteristicas de la Tabla Periodica: Que es??
What are the names of the two elemental groups from the periodic table used in your device? Discuss how the properties of these elements will be important for their function in your innovative device.
The________________group has characteristics including _______________ and __________________. This is important in my device because_____________________.
Submit your Innovative Device application here.
Options: Build a House, A Fuel Efficient Car, Energy Saving Appliance, Solar-powered devices, Alternative Energy Device, Medical Equipment
Element Classes PPT/Notes
In teams: Discovering Pure Materials: Observation and Classification
Common materials include: charcoal, coins, chalk, nails, aluminum, plastics, oil, water, sand/glass, granite, ceramic
Tools: hammer, conductivity meter, hot plate, burner, thermometer, balance, ruler
Identify the main groups you will be observing from the periodic table?
Pick at least one sample from two periodic groups and compare their characteristics.
Put your observations into your Periodic Table Characteristics, Caracteristicas de Tabla Periodica.
Fill in a Class Periodic Table so that our images/videos can be hyperlinked like the TedEd table.
The________________group has characteristics including _______________ and __________________. This is important in my device because_____________________.
Submit your Innovative Device application here.
Options: Build a House, A Fuel Efficient Car, Energy Saving Appliance, Solar-powered devices, Alternative Energy Device, Medical Equipment
Element Classes PPT/Notes
In teams: Discovering Pure Materials: Observation and Classification
Common materials include: charcoal, coins, chalk, nails, aluminum, plastics, oil, water, sand/glass, granite, ceramic
Tools: hammer, conductivity meter, hot plate, burner, thermometer, balance, ruler
Identify the main groups you will be observing from the periodic table?
Pick at least one sample from two periodic groups and compare their characteristics.
Put your observations into your Periodic Table Characteristics, Caracteristicas de Tabla Periodica.
Fill in a Class Periodic Table so that our images/videos can be hyperlinked like the TedEd table.
First, Understanding the Properties of Elements
In the Electrolytic Cell Lab, you will need to be aware of at least four element groups and their properties in order to run a safe and effective lab.
1. Learn vocabulary terms-properties: conductive, insulating, malleable, ductile, brittle, reactive, inert, semi-conductive and electronegative
& energy transformations- efficient, mechanical, electrical, radiant, chemical and electromagnetic.
2. Background reading- Insulation, Activity-Identifying Good Insulators & Insulators PPT (keep safety precaution & efficiency in mind)
3. Read Electrolysis & Measuring Electricity
4. Lab-Electrolytic Cell & Measuring Electricity Also Try Ch 21Activity: Lemon Battery
Extension:
Read your Chemistry Text
- sec 21.1, Electrochemical cells and complete 21.1 assessment.
- share on a google doc titled: Ch21 Electrochemical cells (your name)
Next, Quantify/Qualify Different Energy Forms
You will describe how one form changes into another and quantify/qualify energylost in the transformation.
1. Energy Efficiency Quiz
2. Read Electricity & Electromagnetism
3. Lab-Static Electricity
4. Lab-Electromagnetic Cell
Finally, use your knowledge of atomic ionization energy, electronegativity and radii
Discuss how specific materials are highly functional for a given device.
1. Read Photovoltaic Cells, p2-11.
2. Lab-Photovoltaics: Wiring & Temperature
3. Read H2 Educate p3-13.
4. Lab-H2Cell & Solar Car
Extension:
Read your Chemistry Text
- sec 6.3, Periodic Trends and complete 6.3 assessment.
- share on a google doc titled: Ch6 Periodic Trends (your name)
- Complete the Electron Configuration Worksheet
- Watch the Electrified Pickle and turn in your summary printout.
Using the Scientific Method
Discuss a current environmental, economic or energy issue are you addressing with your innovative device?
NEED Science Energy Information Book NEED Building Science NEED Understanding Climate Change
Electrones de Valencia
Electrones de Valencia
Watch Valence Electrons
For each of the seven periodic groups, indicate the number of electrons, or Lewis dots, available for bonding and the principal levels that they come from.
Valence Electrons & Properties PPT
In teams: Each team will work with an element from a periodic groups II-VI
Follow the valence electron shell pair repulsion (VESPR) rules to determine all possible electron arrangements and bond angles for your team's element.
Share your findings with the class/take notes on other presentations in your electron arrangements worksheet.
Bonding/VESPR Model PPT/Notes eConfiguration
Extension:
Read your Chemistry Text
For each of the seven periodic groups, indicate the number of electrons, or Lewis dots, available for bonding and the principal levels that they come from.
Valence Electrons & Properties PPT
In teams: Each team will work with an element from a periodic groups II-VI
Follow the valence electron shell pair repulsion (VESPR) rules to determine all possible electron arrangements and bond angles for your team's element.
Share your findings with the class/take notes on other presentations in your electron arrangements worksheet.
Bonding/VESPR Model PPT/Notes eConfiguration
Extension:
Read your Chemistry Text
- sec 5.2, Electron Arrangement in Atoms and complete 5.2 assessment.
- share on a google doc titled: Ch5 Electron Arrangements (your name)
- read sec 5.3, Physics & the Quantum Mechanic Model
- Quantum Concepts Worksheet.

Energia Ionizacion
Which properties vary depending on the atom's position in the periodic table? Describe how one of these properties changes across and up the periodic table. Submit here. The Nature of the Electron PPT/Notes Lab-Spectroscopy Lab-Ionization Energy Examine the property of color or ionization energy in this lab. Be sure to work on your compound naming skills as well. Extension: Read your Chemistry Text
Ionization Quiz |
| lab-ionizationenergy.pdf | |
| File Size: | 136 kb |
| File Type: | |
Otras Tendencias
How does size (not mass) compare among C, N, O and F ? Is this trend the same or opposite of ionization energy? Submit your response here.
Lab-Making Molecules (trend: atomic radii)
Use the graph of atomic number versus radii to make paper spheres representing each atom in your molecule. Combine these atoms with the correct angles between them to make the molecule.
How does size (not mass) compare among C, N, O and F ? Is this trend the same or opposite of ionization energy? Submit your response here.
Lab-Making Molecules (trend: atomic radii)
Use the graph of atomic number versus radii to make paper spheres representing each atom in your molecule. Combine these atoms with the correct angles between them to make the molecule.
Vinculacion: Ionicos, Covalentes y Metalicos
Ionic Bonding & Ionic Compound Formulas PPT/Notes
Give the formulas and names for as many molecules as you can.
From what you know about covalent molecules (refer back to carbon chemistry), categorize your molecules as ionic or covalent.
Try Ch 7.1 Inquiry Activity: Properties of Compounds
Extension:
Read your Chemistry Text
Prepare for the Element Properties Quiz
& the Ionic Naming & Bonding Quizzes
Bonding and forms of water pollution
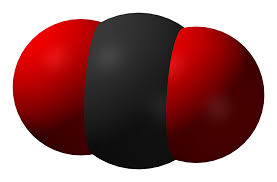
Which of the molecules shown below are ionic and which are covalent?
What are some similarities between the ionic compounds? the covalent compounds?
Metallic Bonding & Intermolecular Forces of Attraction PPT
In teams: Use your Bond Type Organizer to give specific examples of each compound characteristic.
Present an example to the class and take notes on other team examples.
Name three common compounds.
Which atom represents most of the mass of the compound & why? Accurately determine the make-up by mass of one of your compounds. Show your work.
Activity-Heritage Molecule
Percent Composition PPT/Notes
Read your Chemistry Text
& Review Polyatomic Ionic Compounds Quiz
Ionic Bonding & Ionic Compound Formulas PPT/Notes
Give the formulas and names for as many molecules as you can.
From what you know about covalent molecules (refer back to carbon chemistry), categorize your molecules as ionic or covalent.
Try Ch 7.1 Inquiry Activity: Properties of Compounds
Extension:
Read your Chemistry Text
- sec 7.2, Ionic Bonds & Properties and complete 7.1 assessment.
- add this to your google doc titled: Ch7 Forming Ions (your name)
- Complete Ionic Bonding Worksheet
- Complete Covalent Bonding Worksheet
- Memorize the polyatomic ions from the Naming Compounds handout and prepare for the Lab-Chemical Bonding
Prepare for the Element Properties Quiz
& the Ionic Naming & Bonding Quizzes
Bonding and forms of water pollution
Which of the molecules shown below are ionic and which are covalent?
What are some similarities between the ionic compounds? the covalent compounds?
Metallic Bonding & Intermolecular Forces of Attraction PPT
In teams: Use your Bond Type Organizer to give specific examples of each compound characteristic.
Present an example to the class and take notes on other team examples.
Name three common compounds.
Which atom represents most of the mass of the compound & why? Accurately determine the make-up by mass of one of your compounds. Show your work.
Activity-Heritage Molecule
Percent Composition PPT/Notes
Read your Chemistry Text
- sec 10.3, Percent Composition and complete 10.3 assessment.
- submit with your shared Heritage Molecule assignment
- Watch Podcast on Percent Composition & Empirical Formula
- Complete Problem #1 of Empirical Formula 7-4 WS
- Complete Problems 1-3 of Calculating Molecular Formulas WS
- Complete Ionic/Covalent Bond Review Sheet
& Review Polyatomic Ionic Compounds Quiz
| bond_type_organizer.pdf | |
| File Size: | 13 kb |
| File Type: | |
11/4 Lifeline: Feeling Good
Agenda:
Elements & the Periodic Table Guide
Element Boxes
Designer Products
Driving Question
Can we design products that would appeal to a wide range of people and be: economically efficient, visually catchy, chemically tough and durable and most importantly, eco-friendly commodities, so the product must be sustainable for the earth and human kind.
Entry event: Plastic Planet Video
Task 1: Identify the main properties of matter & Role Assignments
Investigating Matter Lab Investigating Alkali Earth Metals Lab
Task 2: Identify Types of Chemical Reactions & Name Compounds
Types of Reactions Lab Making copper Alloys Lab
Writing Reactions
Task 3: Balancing Equations & Product lifecycle
balancing practice I & II
Task 4: Product Design & Final Presentation Rubric (Plastics)/Exam (s)
Agenda:
Elements & the Periodic Table Guide
Element Boxes
Designer Products
Driving Question
Can we design products that would appeal to a wide range of people and be: economically efficient, visually catchy, chemically tough and durable and most importantly, eco-friendly commodities, so the product must be sustainable for the earth and human kind.
Entry event: Plastic Planet Video
Task 1: Identify the main properties of matter & Role Assignments
Investigating Matter Lab Investigating Alkali Earth Metals Lab
Task 2: Identify Types of Chemical Reactions & Name Compounds
Types of Reactions Lab Making copper Alloys Lab
Writing Reactions
Task 3: Balancing Equations & Product lifecycle
balancing practice I & II
Task 4: Product Design & Final Presentation Rubric (Plastics)/Exam (s)