Menu
Oxidation and Reduction
Tendencias del Estado de Oxidacion Practica con Estados de Oxidacion
Estados de la Oxidacion de Oxigeno Extranos Oxidacion y Reduccion
Redox con Hierro Redox con Zinc en Sulfato de Cobre Voltaic Celulas
Estados de la Oxidacion de Oxigeno Extranos Oxidacion y Reduccion
Redox con Hierro Redox con Zinc en Sulfato de Cobre Voltaic Celulas
Oxidation state trends in the periodic table

Group 1 2 5 6 7
H+- Li+ Be2+ N3- O2- F1-
alkali alkaline earth nonmetals halogen
oxidation states
Li+F- H+ O2- Li+ N3-
neutral compound
Li F H2O Li3N
H+- Li+ Be2+ N3- O2- F1-
alkali alkaline earth nonmetals halogen
oxidation states
Li+F- H+ O2- Li+ N3-
neutral compound
Li F H2O Li3N
Watch Khan Academy's video on Oxidation state trends.
For information in Espanol go to: http://www.classzone.com/cz/books/woc_07/page_build.htm?id=resources/jsp/audio_downloads/audio_download4
Practice combining metals and nonmetals using their oxidation states to create a neutral compound.
Practice naming these binary ionic (only 2 ions) compounds.
For information in Espanol go to: http://www.classzone.com/cz/books/woc_07/page_build.htm?id=resources/jsp/audio_downloads/audio_download4
Practice combining metals and nonmetals using their oxidation states to create a neutral compound.
Practice naming these binary ionic (only 2 ions) compounds.
Practice with oxidation states
Watch Khan Academy's video on Practicing oxidation states.
Practice determining the oxidation states of compounds shown above.
Practice naming them. Extra credit for the determining oxidation stated and naming HCO3-.
Practice determining the oxidation states of compounds shown above.
Practice naming them. Extra credit for the determining oxidation stated and naming HCO3-.
Unusual oxygen oxidation states

Elements such as oxygen can change oxidation states depending on the periodic trends such as electronegativity of the element it bonds to.
ie. H-O-O-H or H2O2 The more electronegative oxygens hold a 1- charge each next to the 1+ hydrogens.
F-O-F or OF2 The more electronegative fluorine atoms pull electrons away and oxidize the oxygen atom to have a 2+ charge.
Read EEI’s Lesson 5: Sources That Destroy Ozone.
Answer Part 1 & 2.
Identify three substances that are likely to react easily with ozone.
Be able to draw the structure, give the formula and the ionic charges for each substance.
For instance,
F
F-C-Cl chlorodifluoromethane
H
This HCFC, hydrofluorochlorocarbon, is a newer molecule that is less likely to react with ozone because the added hydrogen is less electronegative. The other atoms which are halogens pull electrons away and oxidize the carbon to a -1 charge.
ie. H-O-O-H or H2O2 The more electronegative oxygens hold a 1- charge each next to the 1+ hydrogens.
F-O-F or OF2 The more electronegative fluorine atoms pull electrons away and oxidize the oxygen atom to have a 2+ charge.
Read EEI’s Lesson 5: Sources That Destroy Ozone.
Answer Part 1 & 2.
Identify three substances that are likely to react easily with ozone.
Be able to draw the structure, give the formula and the ionic charges for each substance.
For instance,
F
F-C-Cl chlorodifluoromethane
H
This HCFC, hydrofluorochlorocarbon, is a newer molecule that is less likely to react with ozone because the added hydrogen is less electronegative. The other atoms which are halogens pull electrons away and oxidize the carbon to a -1 charge.
| lesson-environmentozonehuman_influence_workbook.pdf | |
| File Size: | 536 kb |
| File Type: | |
| assessement-environmentozonehuman_influence.pdf | |
| File Size: | 331 kb |
| File Type: | |
Redox with Iron
Write out the complete balanced equation for the oxidation-reduction reaction of iron sulfide:
Fe + S --> Fe S
What is the charge for Fe initially? finally?
What is the charge for S initially? finally?
Which element is being oxidized? reduced?
Write out the half reactions
Fe --> Fe2+ + 2 e- Fe oxidized
S + ______e- --> S2- S reduced
What about iron rusting in oxygen?
Fe + S --> Fe S
What is the charge for Fe initially? finally?
What is the charge for S initially? finally?
Which element is being oxidized? reduced?
Write out the half reactions
Fe --> Fe2+ + 2 e- Fe oxidized
S + ______e- --> S2- S reduced
What about iron rusting in oxygen?
Write out the complete balanced equation for the oxidation-reduction reaction of iron(III)oxide:
____ Fe + ____O2 --> _____Fe O
What is the charge for Fe initially? finally?
What is the charge for S initially? finally?
Which element is being oxidized? reduced?
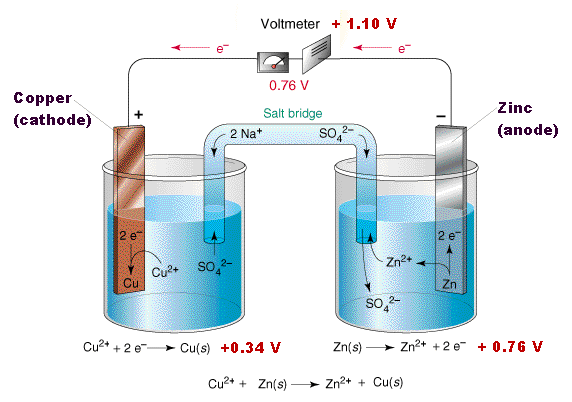
Redox with zinc and coppersulfate
____ Fe + ____O2 --> _____Fe O
What is the charge for Fe initially? finally?
What is the charge for S initially? finally?
Which element is being oxidized? reduced?
Redox with zinc and coppersulfate
Watch Khan Academy's: Redox Reaction from Dissolving Zinc in Copper Sulfate.
Use the electronegativity table above to determine which of the following metals will also become oxidized: Cd, Pd, Fe, Ni and/or Al.
Write out the half reactions for each redox.
For example: Cu has a higher electronegativity than Fe. Fe will be oxidized:
Fe (s) --> Fe 2+ (aq) + 2e-
Cu2+ (aq) + 2e- --> Cu (s) Cu reduced and the aqueous solution will become clear.
SO4 2- does not change oxidation state.
Now, for more practice try the following:
Use the electronegativity table above to determine which of the following metals will also become oxidized: Cd, Pd, Fe, Ni and/or Al.
Write out the half reactions for each redox.
For example: Cu has a higher electronegativity than Fe. Fe will be oxidized:
Fe (s) --> Fe 2+ (aq) + 2e-
Cu2+ (aq) + 2e- --> Cu (s) Cu reduced and the aqueous solution will become clear.
SO4 2- does not change oxidation state.
Now, for more practice try the following:
| worksheet-redox_reactions2013.html | |
| File Size: | 89 kb |
| File Type: | html |
Prepare for Redox Reactions and Electrochemistry Quiz, Zumdahl, 18A: http://www.classzone.com/cz/books/woc_07/quiz.htm?title=Chapter%20Quiz&ch=4&testId=17966
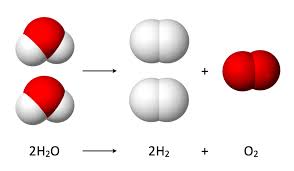
Electrolysis: Reduction and Oxidation of water using electricity
versus
Electrolysis: Reduction and Oxidation of water using electricity
versus
| lesson-electrolysischemiosmosis.pdf | |
| File Size: | 115 kb |
| File Type: | |
Video previewing activity
1. Watch the electrolysis video on classzone.com, Ch 18 videos. http://www.classzone.com/cz/books/woc_07/get_chapter_group.htm?cin=4&rg=ani_chem&at=videos&var=videos
2. Answer and printout the follow-up questions to turn in tomorrow.
more questions to think about....
Why does one buret have twice as much gas as the other?
What are the new oxidation states for hydrogen and oxygen?
Which element is being oxidized, and which is reduced?
Complete each half reaction:
____H2O + _____ --> _____ OH- + _____H2
____H2O --> O2 + ______H+ + _______
What are three places this complete reaction is taking place around you?
1. Watch the electrolysis video on classzone.com, Ch 18 videos. http://www.classzone.com/cz/books/woc_07/get_chapter_group.htm?cin=4&rg=ani_chem&at=videos&var=videos
2. Answer and printout the follow-up questions to turn in tomorrow.
more questions to think about....
Why does one buret have twice as much gas as the other?
What are the new oxidation states for hydrogen and oxygen?
Which element is being oxidized, and which is reduced?
Complete each half reaction:
____H2O + _____ --> _____ OH- + _____H2
____H2O --> O2 + ______H+ + _______
What are three places this complete reaction is taking place around you?
| lab-electrolysis_of_water.pdf | |
| File Size: | 543 kb |
| File Type: | |

Photolysis: Reduction and oxidation of water using light
The Main Points:
1. Chemical reactions are necessary for life.
2. Energy is transferred from one form to another.
(in oxidation-reduction, energy is transferred via electrons)
3. Chemiosmosis is a physical process in living things that transfers electrical
energy to chemical rich energy in this case forming ATP (adenosine triphosphate).
| activity-chemiosmosis-atp_synthesis.pdf | |
| File Size: | 179 kb |
| File Type: | |
| summary-chemiosmosis-atp_formation.pdf | |
| File Size: | 503 kb |
| File Type: | |
| worksheet-modeling_photorespproducts.pdf | |
| File Size: | 157 kb |
| File Type: | |
Using Redox for Future Energy Solutions
Now describe how energy is transferred using the materials in each system.
For example,
In an electrolytic cell, electric energy is transferred via electrons to form hydrogen gas. This gas stores the new energy in its chemical rich bonds.
In a photolytic reaction, _____________ energy is transformed into _______________________electrons. Then, ____________________is oxidized to form oxygen gas.
In a fuel cell, ___________________________________________________________________________________ . _________________________________________forming hydrogen gas for fuel.
In a solar cell, ________________________________________________________________________________. _____________________________________________________________________.
Prepare for Oxidation/Reduction and Balancing Quiz, Zumdahl 18C
http://www.classzone.com/cz/books/woc_07/quiz.htm?title=Chapter%20Quiz&ch=4&testId=17967
For example,
In an electrolytic cell, electric energy is transferred via electrons to form hydrogen gas. This gas stores the new energy in its chemical rich bonds.
In a photolytic reaction, _____________ energy is transformed into _______________________electrons. Then, ____________________is oxidized to form oxygen gas.
In a fuel cell, ___________________________________________________________________________________ . _________________________________________forming hydrogen gas for fuel.
In a solar cell, ________________________________________________________________________________. _____________________________________________________________________.
Prepare for Oxidation/Reduction and Balancing Quiz, Zumdahl 18C
http://www.classzone.com/cz/books/woc_07/quiz.htm?title=Chapter%20Quiz&ch=4&testId=17967
Voltaic cells

Cathode materials
The most common compounds used for cathode materials are LiCoO2, LiNiO2 and LiMn2O4. Of these, LiCoO2 has the best performance but is very high in cost, is toxic and has a limited lithium content range over which it is stable. LiNiO2 is more stable, however the nickel ions can disorder. LiMn2O4 is generally the best value for money, and is also better for the environment.
Anode material
The anode material is carbon based, usually with composition Li0.5C6. This lithium content is lower than would be ideal, however higher capacity carbons pose safety issues.
Electrolyte
Since lithium reacts violently with water, and the cell voltage is so high that water would decompose, a non-aqueous electrolyte must be used. A typical electrolyte is LiPF6 dissolved in an ethylene carbonate and dimethyl carbonate mixture.
The following reactions take place upon discharge:
At the anode: xLi+ + Mn2O4 → LixMn2O4
At the cathode: LixC6→ xLi+ + 6C + xe-
Overall: LixMn2O4 + 6C → LixC6 + Mn2O4
The most common compounds used for cathode materials are LiCoO2, LiNiO2 and LiMn2O4. Of these, LiCoO2 has the best performance but is very high in cost, is toxic and has a limited lithium content range over which it is stable. LiNiO2 is more stable, however the nickel ions can disorder. LiMn2O4 is generally the best value for money, and is also better for the environment.
Anode material
The anode material is carbon based, usually with composition Li0.5C6. This lithium content is lower than would be ideal, however higher capacity carbons pose safety issues.
Electrolyte
Since lithium reacts violently with water, and the cell voltage is so high that water would decompose, a non-aqueous electrolyte must be used. A typical electrolyte is LiPF6 dissolved in an ethylene carbonate and dimethyl carbonate mixture.
The following reactions take place upon discharge:
At the anode: xLi+ + Mn2O4 → LixMn2O4
At the cathode: LixC6→ xLi+ + 6C + xe-
Overall: LixMn2O4 + 6C → LixC6 + Mn2O4
Oxidation state trends in the periodic table Practice with oxidation states Unusual oxygen oxidation states Oxidation/reduction (redox) reactions Redox with Iron Redox with zinc in copper sulfate Voltaic cells